Targeting Inflammation
Targeting inflammation in obese patients is critical for mitigating metabolic dysfunction, reducing comorbid disease risk, and improving treatment outcomes across multiple organ systems.
Obesity is characterized not only by excess adiposity but also by chronic low-grade systemic inflammation driven by adipose tissue-derived cytokines and immune cell infiltration. Adipocytes in obese individuals secrete pro-inflammatory mediators such as TNF-α, IL-6, and MCP-1, which contribute to insulin resistance, endothelial dysfunction, and hepatic steatosis.1,2 This inflammatory milieu disrupts metabolic homeostasis and accelerates the development of type 2 diabetes, cardiovascular disease, non-alcoholic fatty liver disease, IMIDs and others. Multiomics analyses have identified several immune-related genes as key drivers of severe obesity, underscoring the molecular link between inflammation and adiposity.1

Thus, therapeutic strategies that target inflammation—either directly or through weight reduction—can significantly improve clinical outcomes.3 Improving patients’ lifestyle habits, in terms of nutrition (e.g., energy-reduced diet, adjusted macronutrient ratio, etc.) and frequency/type of physical activity, remains a cornerstone of obesity management.4
Medications
Anti-obesity medications (AOMs) have served an increasingly important role in comprehensive obesity treatment and should be administered in combination with lifestyle interventions. Early agents include orlistat, which modifies digestive tract absorption, and centrally acting drugs, such as phentermine-topiramate and naltrexone-bupropion, which regulate appetite in the brain.5
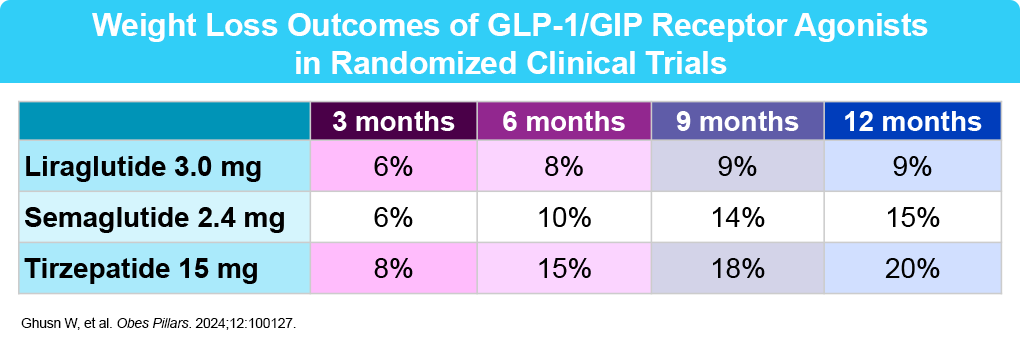
Recently approved anti-obesity medications include glucagon-like peptide-1 (GLP-1) receptor agonists (liraglutide and semaglutide) and a combined GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) agonist (tirzepatide). GLP-1, endogenously produced in cells of the distal ileum and colon, modulates appetite, leading to decreased food intake and consequent weight reduction.6 Centrally, GLP-1 promotes satiety through the activation of GLP-1 receptors in the hypothalamus and brainstem, which causes a reduction in food intake.4 GIP is a nutrient-stimulated hormone that can improve lipid and glucose metabolism.7,8 Overall, these medications work by slowing digestion, reducing appetite, and decreasing cravings. Some, like liraglutide, require daily injections, while the others (semaglutide and tirzepatide) are typically administered weekly.9,10 At this time, there is one GLP-1 daily pill formulation approved by the FDA for weight loss – oral semaglutide – which was approved in December 2025. Orforglipron is another oral agent that is under investigation as a small-molecule, nonpeptide oral GLP-1 RA, with positive Phase III efficacy and safety results. 11
In general, these agents are well tolerated. The side effect profile includes predominantly mild to moderate GI side effects (nausea, diarrhea, constipation, vomiting) which most frequently do not require discontinuation. Serious events like gallbladder disorders or acute pancreatitis may occur, but these are rare. Clinicians need to prioritize individualized dosing and ongoing patient assessment and follow-up to optimize safety and efficacy. Potential new considerations include aspiration risk during anesthesia due to delayed gastric emptying and issues with bowel preparation for colonoscopies. While effective for obesity management, vigilant patient monitoring is critical to address and mitigate potential risks.12
To address the growing global rates of obesity, the World Health Organization (WHO) released its first guideline supporting the use of GLP-1/GIP therapies in December 2025.13 The two guidelines are as follows:
- In adults living with obesity, GLP-1 therapies may be used as a long-term treatment for obesity.
- In adults living with obesity who are prescribed GLP-1 therapies, intensive behavioral therapy may be provided as part of a comprehensive multimodal clinical algorithm.
The Obesity Association, a division of the American Diabetes Association (ADA), released similar but more comprehensive guidelines in January 2026.14 These guidelines align with preferred use of GLP-1 and GLP-1/GIP RAs to manage obesity, and provide several additional recommendations surrounding lifestyle interventions, treatment goals, medications, dosing, comorbidities, special populations, and counseling.
Other interventions for treating obesity include endoscopic and bariatric procedures, and psychotherapy, each of which have roles in overall obesity/inflammation treatment. A combination of these management approaches may lead to immunity equilibrium, beneficial gut microbiome signatures, better overall overweight/obesity control, and prevention and/or management of complications by stopping the overweight/obesity-inflammation cycle.4 By thinking of obesity-related inflammation as a central pathophysiologic mechanism, clinicians can better tailor treatments for their patients and reduce the burden of obesity-related complications.
References
- Chen HH, et al. Cell Genom. 2025;5:100784.
- Jensen MD, et al. Circulation. 2014;129(25 Suppl 2):S102-38.
- Wong HJ, et al. Diabetes Care. 2025;48:292-300.
- Gkrinia EMM, Belančić A. Curr Issues Mol Biol. 2025;47:357.
- Gudzune K, et al. JAMA. 2024;332(7):571-584.
- Kadouh H, et al. J Clin Endocrinol Metab. 2020;105:1552-1563.
- Jastreboff AM, et al. N Engl J Med. 2025;392:958-971.
- Samms R, et al. Trends Endocrinol Metab. 2020;31:410-421.
- Moore PW, et al. Adv Ther. 2023;40:723-742.
- Lilly USA, LLC. Disease Education Resources: Obesity. https://medical.lilly.com/us/diseases/disease-education-resources/obesity/obesity. Accessed 1/7/26.
- Wharton S, et al. NEJM. 2025;393:1796-1806.
- Ghusn W, et al. Obes Pillars. 2024;12:100127.
World Health Organization. WHO guideline on the use of glucagon-like peptide-1 (GLP-1) therapies for the treatment of obesity in adults. 2025. https://app.magicapp.org/#/guideline/LrRxrL. Accessed 1/7/26.
- ADA Professional Practice Committee for Obesity. BMJ Open Diab Res Care. 2026;13:1